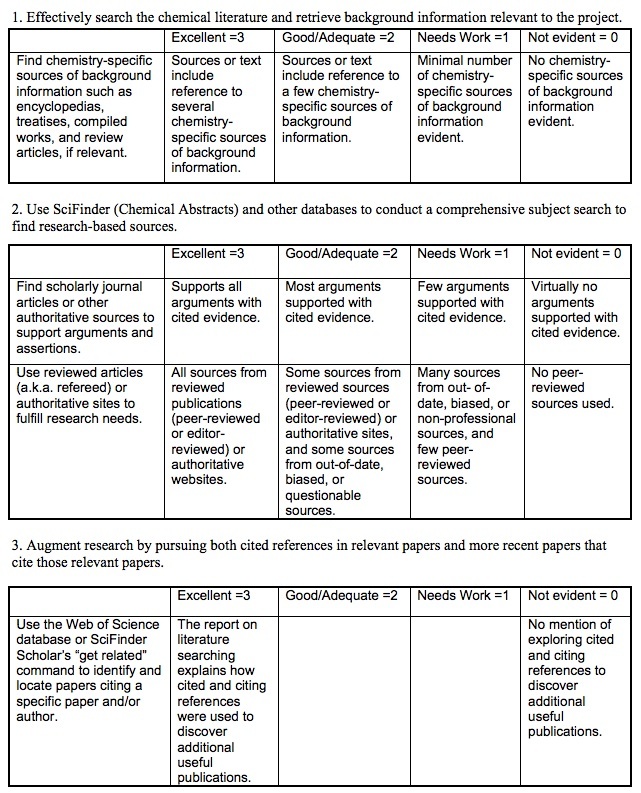
Law of conservation of mass Essay - 773 Words.
Design equations under isothermal conditions are based on mass conservation law.Component concentrations inside a tubular reactor are functions of axial locations, so we will select an infinitesimal volume element, dV r, as the control region (see Fig. 4.2).Based on the plug flow assumption, at steady-state, for any component we can write a mass balance equation for volume dV r.
The law of conservation of mass states that mass can neither be created nor destroyed in a chemical reaction. Thus, the amount of matter cannot change. Sir Antoine Lavoisier promoted this idea. This law says that when a chemical reaction rearranges atoms into a new product, the mass of the reactants (chemicals before the chemical reaction) is the same as the mass of the products (the new.
Laws of Conservation of Mass and Energy 1224 Words 5 Pages Introduction In any science that requires computation, specifically in Chemistry, gathering measurements of various physical and chemical properties need to be strictly accurate and precise for it is in this most crucial phase that either make or brake the quality and value of science.
The Law of Conservation of Mass revolutionized the study of chemistry and is one of its most important principles. Although discovered by multiple researchers, its formulation is most often attributed to French scientist Antoine Lavoisier and is sometimes named after him.
Answer to: Law of conservation of mass examples? By signing up, you'll get thousands of step-by-step solutions to your homework questions. You can.
The conservation of mass. AIM. The aim of this experiment is to show that mass is conserved during a chemical reaction. The Law of Conservation of Mass, established in 1789 by French Chemist Antoine Lavoisier, states that mass is neither created nor destroyed in any ordinary chemical reaction.
Law of Conservation of Mass Examples Combustion process: Burning of wood is a conservation of mass as the burning of wood involves Oxygen, Carbon dioxide, water vapor and ashes. Chemical reactions: To get one molecule of H 2 O (water) with the molecular weight of 10, Hydrogen with molecular weight 2 is added with Oxygen whose molecular weight is 8, thereby conserving the mass.